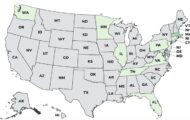
Protica Inc. of Pennsylvania is recalling four of their protein drink products because they may be contaminated with Clostridium botulinum. That bacteria can cause a fatal illness. No illnesses have been confirmed to date.
 You can see the long list of recalled product details at the FDA site. The products that are recalled are Protein to Go Milk Chocolate shake in 2.5 ounce bottles, Protein to Go French Vanilla Latte in 2.5 ounce bottles, Nutritional Resources Protein Wave gelatin in 6 ounce cups, and Body Choice Protein Shots in 3 ounce vials. The Manufacture Dates and Lot Numbers can be found at the FDA web site.
You can see the long list of recalled product details at the FDA site. The products that are recalled are Protein to Go Milk Chocolate shake in 2.5 ounce bottles, Protein to Go French Vanilla Latte in 2.5 ounce bottles, Nutritional Resources Protein Wave gelatin in 6 ounce cups, and Body Choice Protein Shots in 3 ounce vials. The Manufacture Dates and Lot Numbers can be found at the FDA web site.
Botulism poisoning symptoms include double vision, blurred vision, general weakness, dizziness, and trouble speaking or swallowing. If anyone is experiencing these symptoms they should seek immediate medical attention. Botulism poisoning is survivable if treated immediately. For questions, call Protica at 1-800-776-8422.