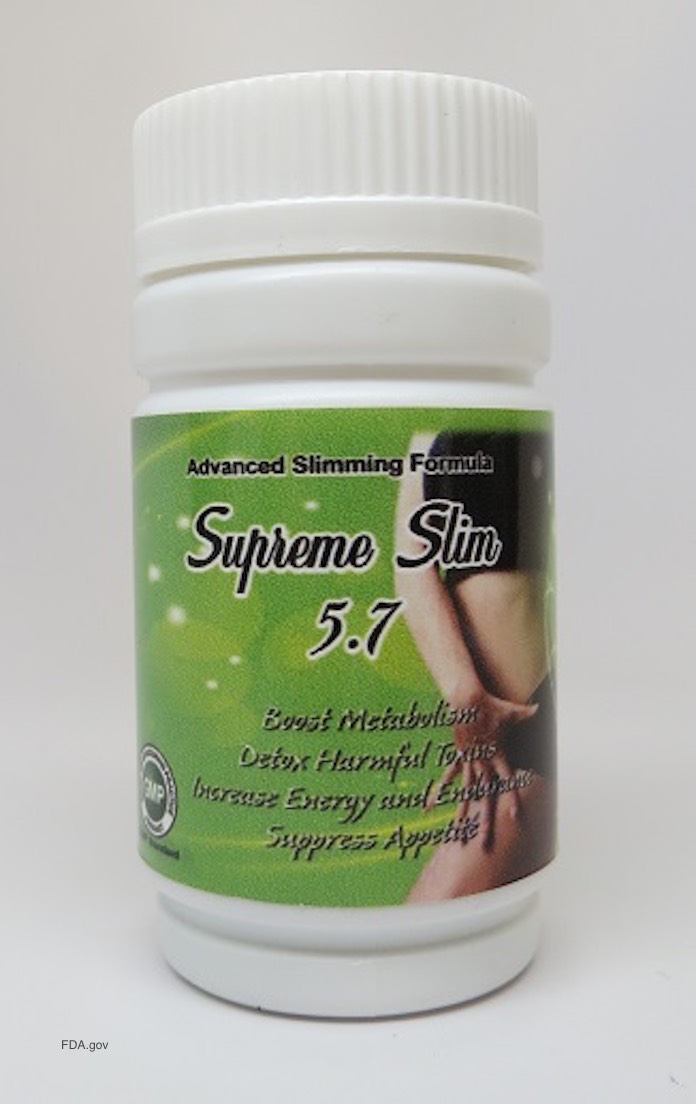
The FDA is warning the public not to purchase or use Supreme Slim 5.7, a dietary supplement that is sold for weight loss on some web sites and may have been sold in retail stores. It contains phenolphthalein and sildenafil, two undeclared drugs.
Phenolphthalein is not approved for use and it may be associated with an increased risk of cancer. Sildenafil is the active ingredient in the prescription drug Viagra. It may interact with nitrates found in some prescription drugs, and can lower blood pressure to dangerous levels.
If you purchased this product, do not consume it. Take it to the drug disposal program in your area or take it back to the place of purchase for a refund.
You can report adverse effects from taking this product or any dietary supplement to the FDA. Use the FDA’s MedWatch Online Reporting Form.
The FDA states that “This notification is to inform the public of a growing trend of dietary supplements or conventional foods with hidden drugs and chemicals. These products are typically promoted for sexual enhancement, weight loss, and body building and are often represented as being “all natural.” FDA is unable to test and identify all products marketed as dietary supplements that have potentially harmful hidden ingredients. Consumers should exercise caution before purchasing any product in the above categories.”