The United States filed a civil complaint against Michel Cordon Bleu Inc. of Los Angeles, California to prevent the distribution of adulterated seafood products on Tuesday, October 3, 2017. This complaint was filed in the US. District Court for the Central District of California. The Court was asked for a permanent injunction to shut down the facility which is located at 3625 South Western Avenue in Los Angeles.

That company prepares, processed, packs, holds, and distributes vacuum-packed, ready-to-eat cold and hot smoked fish and fishery products.
The complaint alleges that Michel Cordon Bleu and Michel G. Blanchet adulterated seafood products by “preparing, packing, or holding them under insanitary conditions whereby the seafood may have become contaminated with filth or may have been rendered injurious to health.” The complaint was filed on behalf of the FDA.
Acting Assistant Attorney General Chad A. Readler of the Justice Department’s Civil Division said in a statement, “The Department of Justice is committed to ensuring that seafood processors comply with laws designed to protect consumers. The Department of Justice will continue to work aggressively with the FDA to ensure that consumers are protected from potentially unsafe food.”
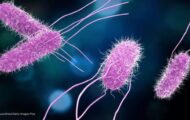
The complaint states that the FDA inspected Michel Cordon Bleu’s facility on many occasions, including twice in 2016. The complaint alleges that the defendants filed to comply with the seafood Hazard Analysis and Critical Control Point (HACCP) regulations by, among other deficiencies, failing to adequately control the risk of Listeria monocytogenes and Clostridium botulinum toxin formation in their vacuum-packed fish or fishery products.
FDA’s testing of environmental samples revealed Listeria monocytogenes in multiple locations throughout the facility. Those samples were collected in January 2016 and in July 2016. The FDA alleges that the defendants did not manufactured, package, and store goods under conditions necessary to minimize the potential for growth of pathogenic bacteria. The defendants allegedly also did not monitor sanitation conditions and practices to make sure they were in conformance with current good manufacturing practices. They also supposedly failed to take corrective action that made sure that the product was not introduced into commerce.
Melinda K. Plaisier, FDA Associate Commissioner for Regulatory Affairs said in a statement, “When we find contaminants that can harm public health at a food manufacturing facility, we must take action to protect consumers. When necessary, we will seek legal action to ensure that manufacturers take steps to comply with food safety laws and regulations.”