Stemvida International Corporation of Ontario, CA is recalling StemAlive capsules sold in 90-count bottles because they contains undeclared milk. People with an allergy or severe sensitivity to milk should not consume this product as they run the risk of serious or life-threatening allergic reaction.
Stemvida International Corporation of Ontario, CA is recalling StemAlive capsules sold in 90-count bottles because they contains undeclared milk. People with an allergy or severe sensitivity to milk should not consume this product as they run the risk of serious or life-threatening allergic reaction.
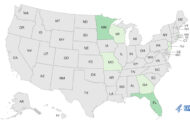
The product was packaged in a white plastic bottle with white lid. The brand name StemVida International appears on the white and gold label. It was distributed to the following states: Arizona, California, Colorado, Florida, New Jersey, New York, Nevada, Oklahoma, Texas and Utah. No illnesses have been reported to date.
The lot number and expiration date is printed in black ink at the bottom of the label. The products under recall have the following lot numbers and expiration dates: 8419 (Exp.05/2015 and 06/2015), 8486 (Exp.07/2015), 8535 (Exp. 08/2015), 8598 (Exp. 10/2015), 8652 (Exp. 12/2015), 8863 (Exp. 01/2016 and 02/2016), 8872 (Exp. 03/2016), 8873 (Exp. 04/2016 and 05/2016), 9040 (Exp. 08/2016 and 09/2016), 9258 (Exp. 10/2016) and 9314 (Exp. 11/2016).
The recall was initiated after it was discovered that the product ingredient list includes bovine colostrum, a known milk allergen but the label did not include an allergen warning.