GlaxoSmithKline (GSK) has recalled all of its Alli weight loss products in the United States, including Puerto Rico, because they think packages may have been tampered with. Some bottles may contain pills that are not Alli capsules. Alli is the only FDA approved over-the-counter weight loss product.
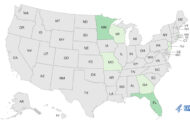
 Consumers in seven states (Alabama, Florida, Louisiana, Mississippi, New York, North Carolina, and Texas) have contacted the company, saying that the Alli bottles they purchased contained “a range of tablets and capsules of various shapes and colors.” Missing labels and fake tamper-evident seals have also been found. Some of the bottles do not have a label. The bottles are in boxes that appear to be authentic packaging.
Consumers in seven states (Alabama, Florida, Louisiana, Mississippi, New York, North Carolina, and Texas) have contacted the company, saying that the Alli bottles they purchased contained “a range of tablets and capsules of various shapes and colors.” Missing labels and fake tamper-evident seals have also been found. Some of the bottles do not have a label. The bottles are in boxes that appear to be authentic packaging.
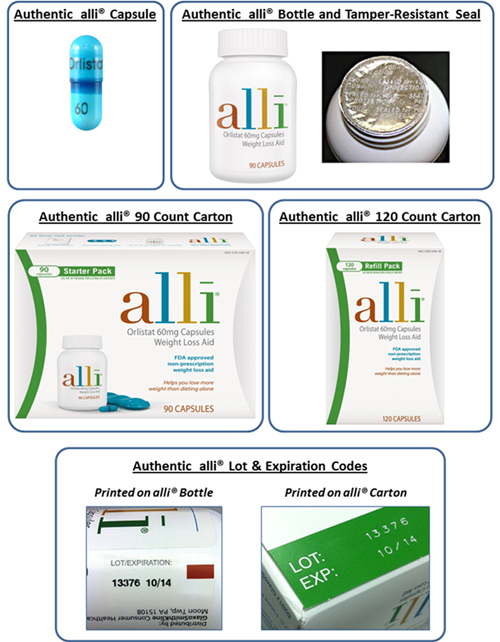
Authentic Alli capsules are turquoise blue with a dark blue band. The capsules are imprinted with “60 Orlistat.” The inner foil seal is imprinted with “Sealed For Your Protection.” The lot numbers and expiration dates of the recalled products are: Carton Lot 14372, Expiration: 02/28/2016; Carton Lot 14395, Expiration: 02/28/2016; Carton Lot 14124, Expiration: 09/30/2015; Carton Lot 14267, Expiration: 01/31/2016; and Carton Lot 14442, Expiration: 04/30/2016.
If any Alli products in your home do not match these descriptions, call the company at 800-671-2554. The company is investigating the matter and is working with the FDA on the recall. If you took a pill that does not match this description, contact your doctor as soon as possible.
Image by GlaxoSmithKline, released for consumer information