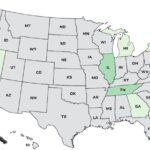
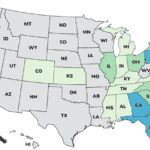
Our top 10 list of 2025 outbreaks continues with the deadly YuShang Listeria monocytogenes outbreak that sickened at least 24 people in nine states and killed three at number eight. Many of the company's products were recalled. The outbreak ended on March 13, 2025. The case count by state is: California (14), Georgia (1), Illinois (2), Michigan (1), New Jersey (1), Nevada (1), New York (1), Oregon (1), and Tennessee (2). Of 24 people who gave information to public health investigators, 22 were hospitalized. The patient age range was less than 1 to 91. Illness onset dates ranged from October 24, 2021 to January 25, 2025. Eight illnesses were related to pregnancy. One pregnant women got sick and recovered. Three unrelated infants were also sick. Two recovered, and one infant who … [Read more...]
Recall of Frozen Raw Oysters From Korea For Norovirus Updated
The recall of frozen raw oysters from Korea for possible norovirus contamination has been updated by the FDA because there are more lot codes, more harvest dates, and another type of frozen oyster included in the warning. The original lot code was B250112 and the frozen raw half shell oysters were harvested on January 12, 2025. There is a norovirus outbreak associated with the consumption of the oysters with that specific lot code. The recall is now for all frozen raw half shell oysters as well as individually quick frozen (IQF) oyster products harvested between December 30, 2024 and February 6, 2025 from Designated Area No. 1 and processed by JBR KR-15-SP. The frozen raw half shell oysters have the lot codes B250103, B250106, B250112, B250114, B250119, B250121, B250123, and … [Read more...]
High Noon Vodka Seltzer Beach Pack Recalled
High Noon Vodka Seltzer Beach Pack is being recalled because it included some Celsius Astro Vibe Energy Drink cans that were inadvertently filled with the vodka seltzer. No illnesses or adverse events have been reported to the company to date in connection with this issue. The recalling firm is High Noon of Modesto, California. Some of the packs contain cans that are filled with the alcoholic vodka seltzer and are mislabeled CELSIUS ASTRO Vibe Energy Drink, Sparkling Blue Razz Edition with a silver top. The labeling error is confined to two lots. The first is High Noon Beach Variety Packs with UPC number 085000040065 and lot codes L CCC 17JL25 14:00 to L CCC 17JL25 23:59 and L CCC 18JL25 00:00 to L CCC 18JL25 03:00. This is a 12 pack with 12 fluid ounce cans with silver … [Read more...]
Bedner Cucumber Salmonella Outbreak Ends with 69 Sick
The Bedner cucumber Salmonella outbreak has ended with 69 people sick in 21 states, according to the CDC. That is an increase of 24 more patients and three more states since the last update was issued on May 30, 2025. The new states are Maryland, Mississippi, and New Jersey. This outbreak is over. The patient case count per state is: Alabama (1), California (1), Colorado (1), Florida (7), Georgia (10), Illinois (5), Indiana (1), Kansas (1), Kentucky (1), Massachusetts (3), Maryland (1), Michigan (2), Mississippi (1), North Carolina (4), New Jersey (3), New York (4), Ohio (6), Pennsylvania (7), South Carolina (6), Tennessee (1), and Virginia (3). Illnesses started on dates ranging from April 2, 2025 to May 29, 2025. The patient age range is from 1 to 89 years. Of the 60 people who … [Read more...]
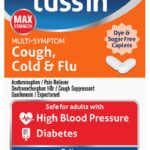
Safetussin Max Strength Blister Packs Recalled For Packaging
Safetussin Max Strength Blister Packs for cough, colds, and flu are being recalled for violating the federal regulations for child resistant packaging. This poses a poisoning risk if children get ahold of the product. No illnesses or injuries have been reported to date in connection with this issue. The distributor is Kramer Laboratories of Bridgewater, New Jersey, a subsidiary of Arcadia Consumer Healthcare of Bridgewater, New Jersey. The product was manufactured in India. This product was sold at HEB, Harris-Teeter, and other regional grocery stores and independently owned pharmacies nationwide from July 2024 through March 2025 for about $11.00. The Safetussin over-the-counter cold medicine contains acetaminophen, which must be in child-resistant packaging as required by the … [Read more...]
Premium Quality Goods Beef Tallow Recalled For No Inspection
Premium Quality Goods Beef Tallow is being recalled for lack of federal inspection. The tallow was processed, packaged, and labeled on behalf of Lady May Tallow by Common Sense Soap of North Walpole, New Hampshire. Common Sense Soap does not possess a federal grant of inspection to produce food products. The tallow contains a nutrition facts label, which may lead people to believe that the product is safe for human consumption, but Common Sense Soap does not have a federal grant of inspection and is not authorized to product products intended for human consumption. No confirmed reports of adverse reactions have been received to date. These products were produced between October 16, 2024 and February 6, 2025. They are all Premium Quality Goods brand. They include: 24 fluid … [Read more...]
Number One Outbreak of 2024 Was Bedner Cucumber Salmonella
The number one outbreak of 2024 was the Bedner cucumber Salmonella outbreak that sickened at least 551 people in 34 states and the District of Columbia. The cucumbers were also sold by Thomas Produce Company. That outbreak ended in August 2024. The case count by state was: Alabama (6), Arkansas (1), California (1), Connecticut (8), Delaware (3), Florida (60), Georgia (48), Illinois (9), Indiana (4), Iowa (5), Kentucky (20), Maine (4), Maryland (17), Massachusetts (11), Michigan (12), Minnesota (10), Mississippi (2), Missouri (4), Nevada (1), New Jersey (22), New York (69), North Carolina (27), Ohio (20), Oklahoma (2), Pennsylvania (68), Rhode Island (8), South Carolina (22), Tennessee (22), Texas (2), Vermont (2), Virginia (48), Washington (1), West Virginia (7), Wisconsin (4), and … [Read more...]
Top 10 2024 Outbreaks: Number Two is WanaBana Lead Poisoning
The top 10 2024 outbreak list continues with number nine: the WanaBana lead illness outbreak. There are at least 500 children sick after eating WanaBana apple sauce pouches, along with Weis cinnamon apple sauce and Schnucks applesauce. The FDA reported 90 children sick, but since these outbreaks are usually much larger than the known cases, we decided to go with the larger CDC number. Each agency used different criteria to identify possible patients. There are 93 confirmed cases of lead poisoning, 233 probable cases, and 28 suspect cases. The patients live in these 41 states: Alabama, Arkansas, California, Colorado, Connecticut, Florida, Georgia, Iowa, Idaho, Illinois, Indiana, Kansas, Kentucky, Louisiana, Massachusetts, Maine, Michigan, Minnesota, Missouri, Mississippi, Montana, … [Read more...]
Top 10 2024 Outbreaks: Number Nine is Milo’s Eggs Salmonella
The top 10 2024 outbreak list continues with the number nine outbreak: the Milo's eggs Salmonella outbreak, which sickened at least 93 people in 12 states. There were 34 people hospitalized because they were so sick. There were several recalls issued in relation to this outbreak. And it's wasn't only chicken eggs that were implicated in this outbreak; duck eggs were included in the recall. The case count by state was: Arizona (1), California (5), Colorado (1), Illinois (12), Iowa (2), Michigan (3), Minnesota (5), New York (1), Oregon (1), Utah (1), Virginia (1), and Wisconsin (60). The patient age range is from 2 to 88 years. Thirty-four people have been hospitalized because they are so sick. That’s a hospitalization rate of 37%, which is much higher than the typical 20% … [Read more...]