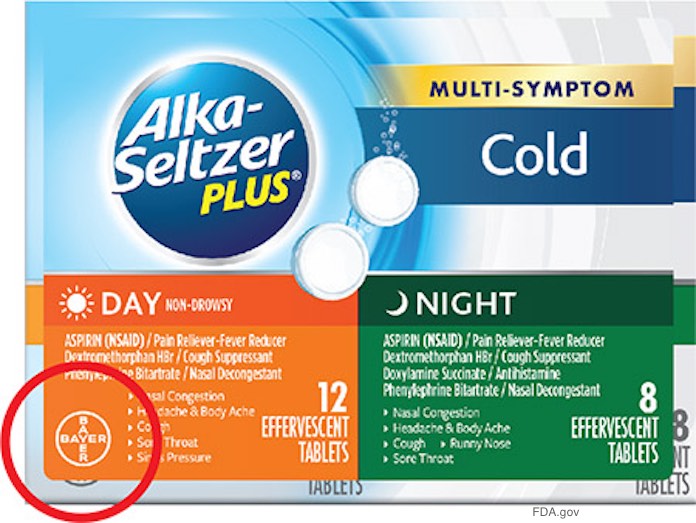
Bayer is voluntarily recalling some Alka Seltzer Plus packages because the ingredients on the front sticker may not match the actual product that is packaged in the carton. These products were sold only in the United States at Walmart, CVS, Walgreens and Kroger (including Dillons Food Stores, Fred Meyer, Fry’s Food Stores, Ralphs, King Soopers and Smith’s Food and Drug) after February 9, 2018.
You can identify the recalled product by checking the Bayer Logo on the lower left corner of the front of the carton. If the logo has an green or orange background, the product is included in the recall. You can see picture off the recalled products at the FDA web site.
The ingredients listed on the front sticker may be different from the ingredients listed on the back of the carton. Consumers may inadvertently ingest a product to which they are allergic, which may cause an anaphylactic reaction. Other consumers may consume an ingredient that is contraindicated for their medical condition or that they may need to avoid. No reports of any adverse health consequences have been reported to the company to date.
These Alka Seltzer products are intended to temporarily relieve symptoms associated with the cold or flu, including congestion, cough, and fever. Bayer is notifying retailers that carried the product electronically and by certified mail. The company is also arranging for returns of all of the recalled products.
If you have purchased one of the containers described above, stop using the product. You can throw away the product, or take it back to the store where you bought it for a refund. You can also report any adverse reactions or quality problems to the FDA’s MedWatch Adverse Event Reporting Program.