There are currently five active investigations on the FDA’s CORE Outbreak Table; four of them have been solved. Four of the outbreaks are caused by various serotypes of Salmonella bacteria. Two of the outbreaks have ended, but the investigations are ongoing.
The first outbreak is a new Salmonella Newport outbreak that has sickened at least 45 people. No food has been identified yet. The FDA has initiated traceback in this outbreak.
The second outbreak is the extensively drug-resistant Salmonella Newport outbreak linked to Rosabella Moringa powder capsules. This is a particularly dangerous outbreak because the drug resistance means it can be much more difficult for doctors to treat serious infections. The case count by state is: Arizona (1), Florida (1), Iowa (1), Illinois (1), Indiana (1), Tennessee (1), and Washington (1). Three people have been hospitalized because they are so sick. Illness onset dates range from November 7, 2025 through January 8, 2026. The patient age range is from 61 to 78 years. While the FDA has said that Ambrosia Brands, the maker of the capsules, should recall all of their moringa powder capsules, they have only agreed to recall certain lots.
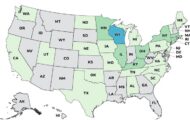
The third outbreak is the Salmonella Typhimurium and Newport outbreak linked to Why Not Natural Organic Moringa Green Superfood capsules and Live It Up Super Greens supplement powders. That outbreak has sickened at least 65 people in 28 states. Fourteen people have been hospitalized. The case count by state is: Alabama (1), Delaware (1), Maine (1), Missouri (1), North Carolina (1), North Dakota (1), New Jersey (1), Oklahoma (1), Pennsylvania (1), Tennessee (1), Texas (1), Utah (1), Washington (1), California (2), Iowa (2), Michigan (2), Nebraska (2), South Carolina (2), Virginia (2), Vermont (2), Connecticut (3), Massachusetts (3), Minnesota (3), Illinois (4), Kentucky (4), Ohio (4), New York (5), and Wisconsin (12). The patient age range is from 3 to 81 years.
The fourth outbreak is a Salmonella Saintpaul outbreak in a not yet identified food that has sickened at least 56 people. The outbreak has ended but the investigation continues. The FDA only initiated traceback.
Finally, the fifth outbreak is the infant botulism outbreak linked to ByHeart powdered infant formula. That outbreak was declared over on March 4, 2026. The case count by state was: The final case count by state is: Arizona (5), California (12), Idaho (2), Illinois (2), Kentucky (1), Massachusetts (2), Michigan (1), Minnesota (3), North Carolina (2), New Jersey (1), Ohio (1), Oregon (3), Pennsylvania (1), Rhode Island (1), Texas (8), Virginia (1), Washington (2), and Wisconsin (1). (This adds up to 50, furthering the confusion.) The infants range in age from 16 to 264 days. Illness onset dates range from December 24, 2023 to November 29, 2025. All of the infants were treated with BabyBIG®, the FDA-approved treatment for this infection developed by the California Department of Health. All of ByHeart’s products have been recalled; if you have any in your home, discard them immediately. If you see them on store shelves, contact the FDA.

If you have been sickened with a food poisoning infection, please contact our experienced attorneys for help with a possible lawsuit at 1-888-377-8900 or text us at 612-261-0856. Our firm represents clients in lawsuits against grocery stores, restaurants, and food processors.