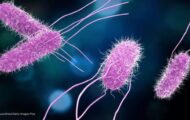
a2 Platinum Premium Infant Formula is being recalled because it may be contaminated with Bacillus cereus, a pathogen that produces the cereulide toxin. No illnesses have been reported to the company to date in connection with the consumption of this product. The recalling firm is the a2 Milk Company (a2MC) of Boulder, Colorado. This formula was distributed nationally through a2MC's website. It was also sold through Amazon and Meijer stores as part of Operation Fly Formula. Importation rights for this product expired on December 31, 2025, and the product has been discontinued and removed from sale. The total number of units is 63,078. About 16,428 were sold to consumers. Cereulide is a heat stable toxin produced bu the Bacillus cereus pathogen. Preparing formula with hot water … [Read more...]
ByHeart Formula Infant Botulism Outbreak Ends With 48 Sick
The ByHeart formula infant botulism outbreak has ended with 48 sick, according to the CDC. This outbreak case count has been confusing from the beginning. In the last update issued on December 11, 2025, there were allegedly 51 babies sick. And the FDA update lists 28 sick. But the FDA numbers include 28 cases of confirmed botulism, 20 cases of probable botulism, and three cases of infants who were eventually diagnosed with another illness. The CDC counts all illnesses except those diagnosed with something else. The last update also included 19 states, but the state count hs been reduced t0 17. One thing remains true: all ByHeart infant formulas were recalled as a result of this outbreak. The final case count by state is: Arizona (5), California (12), Idaho (2), Illinois (2), … [Read more...]
Kendamil Infant Formula Recalled in Canada For Cereulide Toxin
Kendamil Infant Formula is being recalled in Canada because it may be contaminated with cereulide, a toxin produced by Bacillus cereus bacteria that induces vomiting, acute nausea, stomach pain, and sometimes diarrhea. This toxin is heat stable, which means that cooking will not destroy it. No illnesses have been reported to the company to date in connection with the consumption of this infant formula. The recalling firm is Kendal Nutricare Canada Ltd. These products were sold only at Costco Wholesale stores in Canada. This product was sold online and was also sold nationally at the retail level. You can see more pictures of this recalled item at the Canadian Food Inspection Agency web site. The recalled products include Kendamil Infant Formula with Whole Milk - milk based … [Read more...]
ByHeart Infant Formula Recalled For Possible Botulism
ByHeart Infant Formula has been recalled for possible botulism contamination. There is a botulism outbreak that has sickened 13 infants, according to the CDC, that may be associated with this product. The case count of babies with suspected or confirmed infant botulism by state is: Arizona (1), California (2), Illinois (2), Minnesota (1), New Jersey (1), Oregon (1), Pennsylvania (1), Rhode Island (1), Texas (2), and Washington (1). Illness onset dates range from mid-August to November 2025. All 13 infants were treated with BabyBIG®. The infants range in age from 15 to 157 days old. The company has stated, "The FDA has not identified a direct link between any infant formula and these cases and there is no historical precedent of infant formula causing infant botulism." ByHeart … [Read more...]
Perrigo Premium Infant Formula Powder Recalled For High Vit D
Perrigo Premium Infant Formula with Iron Milk Based Powder is being recalled because the levels of vitamin D in the baby formula is above the maximum level permitted. The formula is sold under the brand names CVS Health and HEB Baby. No reports of adverse reactions have been received by the company to date in connection with the consumption of this product. The recalling firm is Perrigo Company of Dublin, Ireland and Allegan, Michigan. The company is recalling three lots within one batch, or 16,500 cans of Perrigo Premium Infant Formula with Iron Milk-Based Powder. It was shipped to HEB Grocery Company in Texas, and to CVS stores in these states: Texas, Florida, California, South Carolina, Virginia, Indiana, Tennessee, New Jersey, Michigan, Pennsylvania, Rhode Island, and Missouri. … [Read more...]
Recall of Crecelac Infant Formula For Cronobacter Updated
The recall of Crecelac Infant Formula for possible Cronobacter contamination has been updated to include more identifying information. There have been no reports of illness in association with the consumption of this product received by the FDA or the company to date. The recalling firm is Dairy Manufacturers Inc. of Prosper, Texas. The recalled product is Crecelac Infant Formula 0-12 months. It is a powdered goat milk infant formula. The UPC number that is stamped on the product label is 8 50042 40847. The lot code for this product is 24 039 1 CHE 352-1 that is printed on the bottom of the can. And the expiration date for this item is 08/2025. Only that lot code had samples containing the Cronobacter bacteria. The containers were distributed mostly in March, April, and May 2024. … [Read more...]
Crecelac Goat Milk Infant Formula Poses Cronobacter Risk
Crecelac Goat Milk Infant Powdered Formula poses a Cronobacter risk according to the FDA. This product was sold at some retail stores in Texas and may have been sold in other locations in the United States. The recalled products from an earlier recall include: CRECELAC INFANT Powdered Goat-Milk Infant Formula with Iron 0 to 12 months - Net Wt. 12.4 ounce (352 grams) Farmalac BABY Powdered Infant Formula with Iron 0 to 12 months Net Wt. 12.4 ounce (352 grams) Farmalac BABY Powdered Infant Formula with Iron Low Lactose 0 to 12 months Net Wt. 12.4 ounce (352 grams) You can see pictures of the recalled products at the FDA web site. Cronobacter was only found in the Crecelac infant formula product. The company did issue a recall of these products on May 24, 2024 because … [Read more...]
Crecelac and Farmalac Infant Formula Recalled For Non-Compliance
Crecelac and Farmalac infant formula is being recalled because it is in non-compliance with the FDA under section 21 CFR 106.110 New Infant formula registration. There have been no reports of injuries or adverse reactions received to date in connection to the consumption of these items. The recalling firm is Dairy Manufacturers Inc. of Prosper, Texas. These products were sold in the United States without complying with FDA's infant formula regulations. The firm had not submitted the required premarket notification. Parents and caregivers shold know that these products have not been evaluated by the FDA to determine whether they meet U.S. food safety and nutrition standards. The products are labeled as infant formula and are packaged in a cardboard and aluminum can of 12.4 ounces … [Read more...]
Enfamil Nutramigen A+ LGG Infant Formula Recalled in Canada
Enfamil Nutramigen A+ LGG Infant Formula is being recalled in Canada for possible Cronobacter contamination. No illnesses have been reported to the company to date in connection with the consumption of the product. The recalling firm is Mead Johnson Nutrition (Canada) Company. Food contaminated with cronobacter can cause serious illness in infants. It is associated with necrotizing enterocolitis and blood poisoning (sepsis), especially in newborns. This product was sold nationally at the retail level. The recalled product is Enfamil Nutramigen A+ LCC Hypoallergenic infant formula that is sold in 561 gram cans. The UPC number that is stamped on the label is 0 56796 00498 2. The item number for the product is 3230626. The expiration date is 01-JA-2025. And the batch number for the … [Read more...]
Nutramigen Hypoallergenic Infant Formula Recalled For Cronobacter
Nutramigen Hypoallergenic Infant Formula is being voluntarily recalled for possible Cronobacter contamination. No illnesses have been reported to the company to date in connection with the consumption of this product. The recalling firm is Reckitt/Mead Johnson Nutrition. The brand name is Enfamil. Cronobacter can cause severe, life-threatening infections or meningitis in infants. Symptoms of this infection can include poor feeding, temperature changes, irritability, jaundice, grunting breaths and abnormal movements. The recalled product is Nutramigen Hypoallergenic Infant Formula powder that is packaged in 12.6 and 19.8 ounce cans. The batch numbers of this recalled infant formula are ZL3FRW, ZL3FPE, ZL3FXJ, ZL3FQD, ZL3FMH, and ZL3FHG. The UPC number is 300871239418 or … [Read more...]