Nicotine pouch makers are being asked by the FDA to use child resistant packaging. The number of nicotine exposure cases reported to U.S. Poison Control Centers have steadily increased from April 1, 2022 to March 31, 2025, according to public health officials. These pouches contain concentrated nicotine that can be harmful or even fatal to young children, even when consumed in small amounts. Doses as low as 1 to 4 milligrams have caused toxic effects in young children. Symptoms of nicotine poisoning can include vomiting, confusion, and loss of consciousness. FDA Commissioner Marty Makary, M.D., M.P.H. said in a statement, "I am concerned about rising reports of nicotine exposures in young children caused by nicotine pouches. The fruity flavors and bright, colorful designs of … [Read more...]
Georgia Pine Gum Spirits of Turpentine Recalled For Packaging
Georgia Pine Gum Spirits of Turpentine is being recalled for insufficient packaging. Turpentine must be in child resistant packaging as required by the Poison Prevention Packaging Act. This product poses a risk of serious injury or death from poisoning if the contents are ingested by young children. No illnesses have been reported to the company to date in connection with this problem. The recalling firm is Diamond G. Forest Products of Patterson, Georgia. About 20,000 units of this product were sold. The recall is for Georgia Pine Gum Spirits of Turpentine. The turpentine is packaged in a clear glass 4 ounce round bottle with a white label and silver top. The Diamond G. Forest Products label is on the front. This product was sold at the retail level at Nature’s Corner Market … [Read more...]
iHerb California Gold Nutrition Iron Supplements Recalled
iHerb California Gold Nutrition Iron Supplements are being recalled because the packaging violates the federal standard for child resistant packaging. No reports of illness have been reported to the company to date in connection with this issue. The recalling firm is iHerb LLC, doing business as Madre Labs LLC, of Irvine, California. The recalled products include California Gold Nutrition Iron Supplements: Daily Prenatal Multi, Ultamins Women’s Multivitamin, and Ultamins Women’s 50+ Multivitamin. They contain iron, which must be in child-resistant packaging as required by the Poison Prevention Packaging Act. The bottles and blister packs are not child proof. The California Gold Nutrition Daily Prenatal Multivitamin bottles are white with a white lid and a gold border label. The … [Read more...]
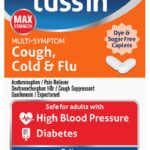
Safetussin Max Strength Blister Packs Recalled For Packaging
Safetussin Max Strength Blister Packs for cough, colds, and flu are being recalled for violating the federal regulations for child resistant packaging. This poses a poisoning risk if children get ahold of the product. No illnesses or injuries have been reported to date in connection with this issue. The distributor is Kramer Laboratories of Bridgewater, New Jersey, a subsidiary of Arcadia Consumer Healthcare of Bridgewater, New Jersey. The product was manufactured in India. This product was sold at HEB, Harris-Teeter, and other regional grocery stores and independently owned pharmacies nationwide from July 2024 through March 2025 for about $11.00. The Safetussin over-the-counter cold medicine contains acetaminophen, which must be in child-resistant packaging as required by the … [Read more...]
NFH Iron Dietary Supplements Recalled For Poisoning Risk
NFH Iron Dietary Supplements are being recalled for poisoning risk because the bottles violate the federal requirement for child resistant packaging. No illnesses or injuries have been reported to the company to date in connection with this problem. The recalling firm is Nutritional Fundamentals for Health (doing business as NFH) of Canada. The recalled dietary supplements are NFH Iron SAP, NFH Heme Iron SAP and NFH Prenatal SAP supplements. They contain iron, which must be in child-resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging is not child-resistant, which pose a risk of poisoning. About 17,660 bottles of this product are included in this recall. The recalled products include Iron SAP, which is Iron Glycinate, in 80 capsules and 12 … [Read more...]