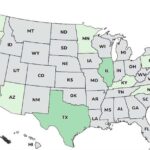
The FDA weighs in on the ByHeart Whole Nutrition Formula botulism outbreak, stating that, "Epidemiologic and laboratory data show that ByHeart Whole Nutrition infant formula might be contaminated with Clostridium botulinum, which is causing infant illness in multiple regions of the country." The case count by state is: Arizona (1), California (2), Illinois (2), Kentucky (1), Minnesota (1), North Carolina (1), New Jersey (1), Oregon (1), Pennsylvania (1), Rhode Island (1), Texas (2), and Washington (1). For 14 infants whose age is available, they range in age from 16 to 157 days. Illness onset dates range from August 9 to November 10, 2025. All of the infants were hospitalized and treated with BabyBIG®, the FDA-approved treatment for this infection developed by the California … [Read more...]
FDA Warned ByHeart About Insanitary Conditions Two Years Ago
The FDA warned ByHeart about insanitary conditions in their facility more than two years ago during the worry about Cronobacter in infant formula. A warning letter was sent on August 30, 2023. The letter states, "During our inspection at the (b)(4) facility, FDA investigators found significant violations of Title 21, Code of Federal Regulations, Part 106 (21 C.F.R. Part 106), Infant Formula Requirements Pertaining to Current Good Manufacturing Practice, Quality Control Procedures, Quality Factors, Records and Reports, and Notifications (“the Infant Formula Rule”)." This information is interesting because powdered ByHeart Whole Nutrition Infant Formula is linked to a botulism outbreak that has sickened 15 infants in 12 states. The California Department of Health conducted testing on … [Read more...]
Minnesota Infant Sickened in ByHeart Formula Botulism Outbreak
A Minnesota infant is sickened in the ByHeart infant formula botulism outbreak, according to the Minnesota Department of Health (MDH). That infant got sick in August 2025. All of ByHeart's formula has been recalled after testing conducted in California found the possible presence of the Clostridium botulinum bacteria in an opened can of that product from the home of a sick infant. In all, there are now 15 infants who are ill after being fed ByHeart Whole Nutrition Infant Formula. The case count by state is: Arizona (1), California (2), Illinois (2), Kentucky (1), Minnesota (1), North Carolina (1), New Jersey (1), Oregon (1), Pennsylvania (1), Rhode Island (1), Texas (2), and Washington (1). For 14 infants whose age was reported to public health officials, they range in age from 16 … [Read more...]
ByHeart Infant Formula Botulism Outbreak Sickens 15 Babies
The ByHeart Infant Formula botulism outbreak has now sickened 15 babies in 12 states, according to the Centers for Disease Control and Prevention (CDC). That is an increase of two more patients in two more states since the last update was issued three days ago. The new states are Kentucky and New Jersey. The case count by state is: Arizona (1), California (2), Illinois (2), Kentucky (1), Minnesota (1), North Carolina (1), New Jersey (1), Oregon (1), Pennsylvania (1), Rhode Island (1), Texas (2), and Washington (1). For 14 infants whose age is available, they range in age from 16 to 157 days. Illness onset dates range from August 9 to November 10, 2025. All of the infants were hospitalized and treated with BabyBIG®. No deaths have been reported. According to information from the … [Read more...]
All ByHeart Infant Formula Is Recalled in Infant Botulism Outbreak
All ByHeart Infant Formula is recalled in relation to the infant botulism outbreak that has now sickened at least 15 babies nationwide. The original recall of two lots of the formula was expanded when the FDA learned that some sick infants had been fed other lots of that product. The California Department of Public Health conducted tests on an opened can of ByHeart Whole Nutrition Infant formula that was fed to and infant who contracted a botulism infection, and preliminary tests suggest the product contained Clostridium botulinum bacteria. The company maintains that no unopened can of their product has tested positive. The FDA is waiting for confirmed results of its testing, but in the meantime ByHeart is recalling all product from the market, and is testing every batch of … [Read more...]
Preliminary Test Suggest Clostridium Botulinum in ByHeart Formula
California preliminary tests suggest that there is Clostridium botulinum in ByHeart infant formula, according to the California Department of Public Health (CDPH). Thirteen babies are sick with botulism poisoning after consuming that formula. The California notice now states that those illnesses are linked to ByHeart Whole Nutrition Infant Formula. ByHeart recalled two lots of that product on November 8, 2025. The product is packaged in a 24 ounce can. The lot numbers and use by dates for the recalled product are Lot: 206VABP/251261P2 (“Use by 01 Dec 2026”) and Lot: 206VABP/251131P2 (“Use by 01 Dec 2026”). This product was sold through the company's website and through retail stores nationwide, including Amazon, Kroger, Walmart, Whole Foods, Target, Sams Club, and others. The … [Read more...]
ByHeart Infant Formula Recalled For Possible Botulism
ByHeart Infant Formula has been recalled for possible botulism contamination. There is a botulism outbreak that has sickened 13 infants, according to the CDC, that may be associated with this product. The case count of babies with suspected or confirmed infant botulism by state is: Arizona (1), California (2), Illinois (2), Minnesota (1), New Jersey (1), Oregon (1), Pennsylvania (1), Rhode Island (1), Texas (2), and Washington (1). Illness onset dates range from mid-August to November 2025. All 13 infants were treated with BabyBIG®. The infants range in age from 15 to 157 days old. The company has stated, "The FDA has not identified a direct link between any infant formula and these cases and there is no historical precedent of infant formula causing infant botulism." ByHeart … [Read more...]
Possible ByHeart Infant Formula Botulism Outbreak Sickens 13
A possible ByHeart infant formula botulism outbreak has sickened 13 babies, according to the Center for Disease Control and Prevention (CDC). All 13 infants have been hospitalized. Two lots of the company's infant formula have been recalled. The formula was sold online and nationwide at the retail level. The case count of babies with suspected or confirmed infant botulism by state is: Arizona (1), California (2), Illinois (2), Minnesota (1), New Jersey (1), Oregon (1), Pennsylvania (1), Rhode Island (1), Texas (2), and Washington (1). Illness onset dates range from mid-August to November 2025. All 13 infants were treated with BabyBIG®. The infants range in age from 15 to 157 days old. Public health officials have been interviewing caregivers about the foods these babies were fed … [Read more...]